CRISPR-integrated gene editing delivery devices market accelerates as biopharma shifts from viral workflows to scalable non-viral systems, driving demand for automated, clinical-grade cell engineering platforms
NEWARK, DE / ACCESS Newswire / March 19, 2026 / According to analysis by Future Market Insights (FMI), the global CRISPR-integrated gene editing delivery devices market is entering a high-growth phase as biopharmaceutical manufacturers transition from research-scale transfection workflows to fully automated, clinical-scale delivery systems capable of supporting commercial cell therapy production.
The market was valued at USD 1.03 billion in 2025 and is projected to reach USD 1.18 billion in 2026. Over the forecast period, demand is expected to expand at a CAGR of 14.10%, ultimately reaching USD 4.41 billion by 2036.
This growth reflects a structural shift in gene therapy manufacturing, where delivery technologies are evolving from supportive tools into critical infrastructure enabling scalable, cost-efficient, and regulatory-compliant genomic medicine production.
Get Access of Report Sample: https://www.futuremarketinsights.com/reports/sample/rep-gb-32375
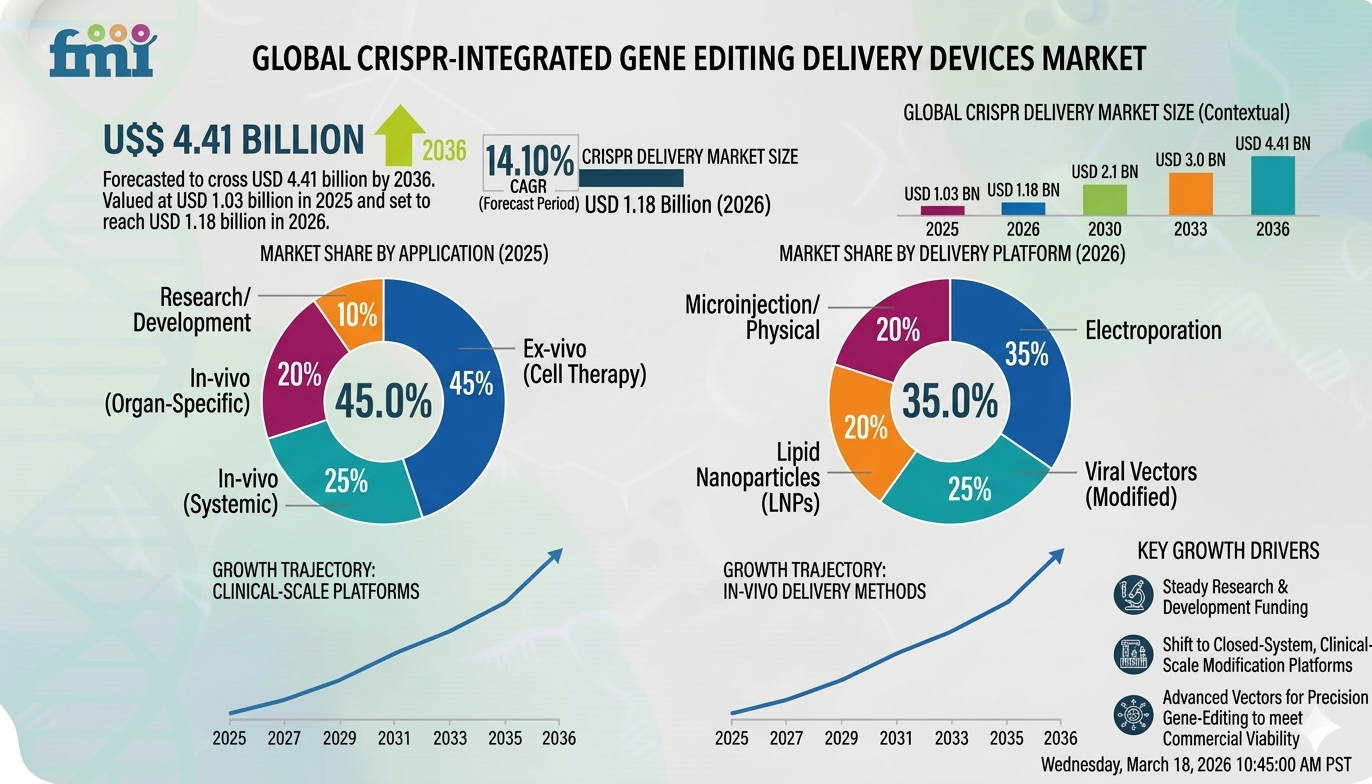
CRISPR-Integrated Gene Editing Delivery Devices Market Snapshot
Market Value (2025): USD 1.03 Billion
Forecast Value (2036): USD 4.41 Billion
Forecast CAGR (2026-2036): 14.10%
Leading Delivery Platform: Electroporation Systems (34.0%)
Top Editing Setting: Ex Vivo Cell Engineering (56.0%)
Key End User: Biopharma & Cell/Gene Therapy Developers (41.0%)
Fastest Growing Market: China
Transition from Viral to Non-Viral Systems Reshaping Manufacturing Strategy
A major driver of the CRISPR delivery devices market is the growing limitation of viral vector-based delivery systems in large-scale manufacturing. While viral transduction has historically enabled gene transfer, it introduces constraints in throughput, cost, and batch consistency.
Biopharmaceutical manufacturing leaders are now faced with a critical decision: continue scaling legacy viral systems or transition toward non-viral delivery architectures such as electroporation and microfluidic platforms.
Non-viral methods are rapidly gaining traction as they:
Eliminate batch-to-batch variability
Enable higher throughput processing
Reduce cost-of-goods for cell therapies
Support multiplex gene editing requirements
As a result, the market is witnessing accelerated adoption of continuous-flow electroporation and automated delivery systems.
Manufacturing Bottlenecks Driving Device Innovation
Autologous and allogeneic cell therapy pipelines are placing unprecedented pressure on delivery technologies. Legacy batch transfection workflows are proving inadequate for commercial-scale production.
Key operational challenges include:
Low-yield transduction limiting scalability
Manual intervention increasing variability
Inability to process large cell volumes efficiently
To overcome these constraints, manufacturers are investing in closed-system, high-throughput delivery platforms capable of processing billions of cells in GMP-compliant environments.
FMI analysts note that companies delaying this transition risk losing competitive advantage due to higher production costs and slower turnaround times.
Electroporation Systems Lead the Market
By delivery platform type, electroporation and flow electroporation systems are expected to account for 34.0% of the market share in 2026.
Their dominance is driven by their ability to provide:
Scalable non-viral delivery pathways
High cell viability and consistency
Compatibility with continuous-flow manufacturing
Unlike chemical transfection or viral methods, electroporation enables reliable, repeatable gene editing at clinical scale, making it the preferred choice for advanced therapy manufacturing.
CRISPR RNP Delivery Gains Momentum
By payload format, CRISPR ribonucleoprotein (RNP) delivery is emerging as a dominant segment due to its transient expression profile and reduced off-target risks.
Key advantages include:
Immediate editing upon cell entry
Lower toxicity compared to DNA-based methods
Reduced risk of unintended genomic alterations
This shift is driving demand for delivery devices specifically optimized to handle sensitive protein-RNA complexes without compromising cell viability.
Ex Vivo Engineering Dominates Clinical Applications
By editing setting, ex vivo cell engineering is projected to hold 56.0% of the market share in 2026.
This dominance is attributed to:
Controlled editing environments outside the human body
Ability to verify genetic modifications before reinfusion
Compliance with stringent regulatory requirements
Ex vivo systems allow manufacturers to ensure quality, safety, and precision, making them the backbone of current clinical gene therapy workflows.
Biopharma Companies Lead Capital Deployment
Biopharma and cell & gene therapy developers are expected to account for 41.0% of total demand in 2026.
These organizations are increasingly:
Internalizing manufacturing capabilities
Investing in proprietary delivery platforms
Reducing reliance on external CDMOs
This shift toward vertical integration is enabling tighter control over production timelines, quality standards, and intellectual property.
Asia-Pacific Emerges as High-Growth Hub
The CRISPR-integrated gene editing delivery devices market is witnessing strong regional growth, particularly in Asia-Pacific, driven by rapid expansion of biotechnology infrastructure.
CRISPR Delivery Devices Market Growth by Country (2026-2036)
China: 15.7%
India: 15.1%
South Korea: 14.8%
United States: 13.8%
United Kingdom: 13.2%
Germany: 12.9%
Japan: 12.4%
China leads global growth due to aggressive investments in translational infrastructure and non-viral delivery systems. India follows closely, supported by expanding biotechnology parks and increasing participation in global clinical trials.
Automation and Continuous-Flow Systems Unlock New Opportunities
Technological advancements are redefining the competitive landscape of CRISPR delivery devices. Continuous-flow electroporation and integrated closed systems are enabling seamless, end-to-end cell engineering workflows.
Emerging innovations include:
Automated delivery platforms integrating isolation, editing, and expansion
Microfluidic systems for precise, low-damage delivery
Lipid nanoparticle frameworks for in vivo applications
These technologies are transforming delivery devices from standalone tools into integrated manufacturing ecosystems.
Key Players in the CRISPR-Integrated Gene Editing Delivery Devices Market
Leading companies operating in the global market include:
MaxCyte, Inc.
Lonza Group AG
Thermo Fisher Scientific Inc.
Bio-Rad Laboratories, Inc.
Cytiva
Takara Bio Inc.
Miltenyi Biotec B.V. & Co. KG
These players are leveraging regulatory expertise, advanced hardware platforms, and proprietary consumables to maintain strong market positioning.
Market Outlook: Delivery Technologies Become Core to Gene Therapy Commercialization
The long-term outlook for the CRISPR-integrated gene editing delivery devices market remains highly positive as gene therapy pipelines continue to expand globally.
With increasing demand for scalable, cost-efficient, and precise gene editing solutions, delivery technologies are becoming central to commercial viability.
As innovation accelerates in automation, non-viral delivery, and integrated manufacturing systems, the market is set to witness sustained growth through 2036-positioning delivery devices as a cornerstone of next-generation genomic medicine infrastructure.
For more insights into the CRISPR-Integrated Gene Editing Delivery Devices Market, visit: https://www.futuremarketinsights.com/reports/crispr-integrated-gene-editing-delivery-devices-market
Related Reports:
Gene Editing Tool Market: https://www.futuremarketinsights.com/reports/gene-editing-tools-market
Microbial Gene Editing Services Market: https://www.futuremarketinsights.com/reports/microbial-gene-editing-services-market
CRISPR-based Gene Editing Market: https://www.futuremarketinsights.com/reports/crispr-based-gene-editing-market
Next Generation Optical Biometry Devices Market: https://www.futuremarketinsights.com/reports/next-generation-optical-biometry-devices-market
Subcutaneous Drug Delivery Devices Market: https://www.futuremarketinsights.com/reports/subcutaneous-drug-delivery-devices-market-share-analysis
About Future Market Insights (FMI)
Future Market Insights (FMI) is a leading provider of market intelligence and consulting services, serving clients in over 150 countries. Headquartered in Delaware, USA, with a global delivery center in India and offices in the UK and UAE, FMI delivers actionable insights to businesses across industries including automotive, technology, consumer products, manufacturing, energy, and chemicals.
An ESOMAR-certified research organization, FMI provides custom and syndicated market reports and consulting services, supporting both Fortune 1,000 companies and SMEs. Its team of 300+ experienced analysts ensures credible, data-driven insights to help clients navigate global markets and identify growth opportunities.
For Press & Corporate Inquiries
Rahul Singh
AVP - Marketing and Growth Strategy
Future Market Insights, Inc.
+91 8600020075
For Sales - sales@futuremarketinsights.com
For Media - Rahul.singh@futuremarketinsights.com
For web - https://www.futuremarketinsights.com
SOURCE: Future Market Insights, Inc.
View the original press release on ACCESS Newswire





